In Ecuador, one member in our party developed significant swelling of her hands and feet.
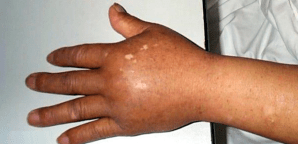
Hand edema
By itself, edema of the hands is not a problem, and it does not require treatment or descent to lower altitudes. But we have mentioned a few other instances in which edema is a big problem – when it occurs in the brain or the lung. In both instances, swelling and fluid leakage happens in response to the low oxygen partial pressure at very high altitude.
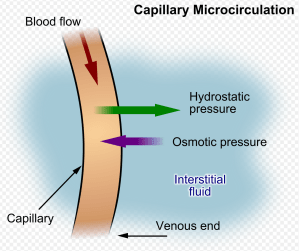
Capillary leak (adapted from unrestricted Wikipedia image)
Is edema advantageous (adaptive) in when it happens in high altitude? The answer is an unequivocal no. The physiology that results in edema is harmful at altitude, but the regulatory mechanisms responsible are adaptive in more common contexts. Here it is important to consider the environment in which a trait evolved. For most of us, this means low altitude. Regulatory traits responsible for edema evolved at low altitudes and operated under different circumstances than Everest mountaineers. The genes responsible for blood vessel regulation did not evolve at 18,000 feet elevation. When we travel to those elevations, things can, and do, go wrong. This is a classic example of gene-environment mismatch.
But do they go right at low altitude. I argue that they do. In fact, we cannot understand why things go wrong at high altitude without understanding how they go right at sea level. To recap:
Hypoxic vasoconstriction in the lung is often useful during pneumonia. But it can cause high altitude pulmonary edema in mountaineers. In combination, high altitude residence and lung infection cause even greater pulmonary edema than would otherwise happen.
Hypoxic cerebral vasodilation at sea level helps the brain cope with severe exertion, trauma, and stroke, compensating by increasing blood flow and oxygen delivery to the brain. The same phenomenon at altitude kills mountaineers with brain edema.
Is edema itself ever really helpful, or is it a side effect?
Edema is a hallmark feature of inflammation. Inflammation, of course, is a key feature of the immune response necessary for the clearance of infection or invasive pathogens. Andy Brainard and I proposed that tissue edema is important for pathogen trapping, an idea we termed the Hemostatic Containment hypothesis. The idea is that leaky vessels do two things. First, leakiness lets white blood cells migrate from blood vessels to the site of infection. Second – the resulting edema expedites pathogen trapping in the microcirculation. The novel idea here is that edema can be a helpful part of the inflammatory response that helps clear infection.
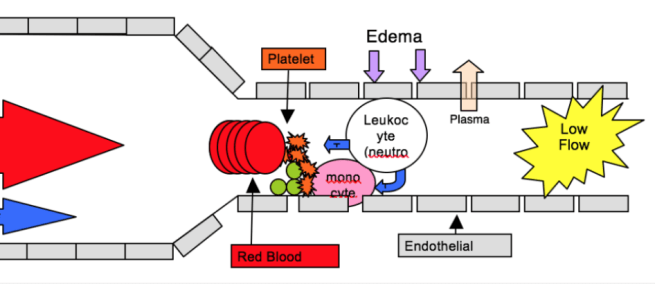
Capillary flow (from left to right) slows after wounding and infection. Plasma loss increases viscosity of blood. Edema reduces capillary flow that allows for pathogen trapping in the microcirculation. Brainard and Alcock termed this phenomenon “Hemostatic Containment”
The hemostatic containment hypothesis predicts that hypoxia-induced inflammation and edema is useful in wounds (a common occurrence for lowlanders). Brainard and Alcock suggested that hemostatic containment is regulated by host factors (the most important being hypoxia), pathogen factors (including bacterial products like lipopolysaccharide), and markers of injury or damage (collagen or tissue factor in the blood).
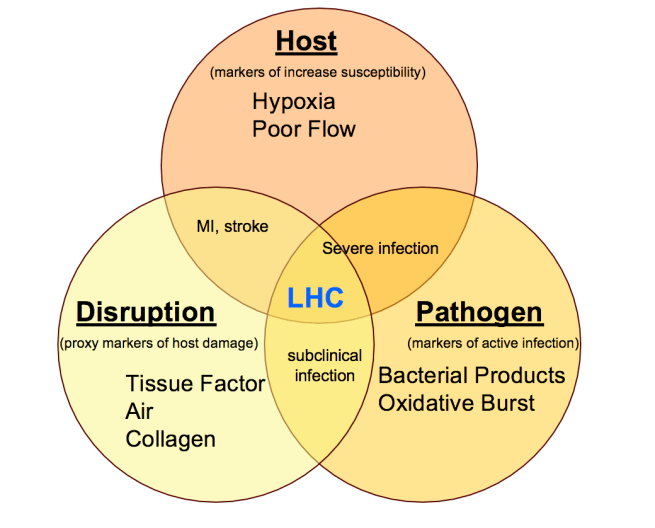
Hemostatic containment is regulated by host factors, pathogen factors, and markers of tissue injury. Hemostatic containment was illustrated in this graphic as “LHC” or leukocyte-assisted hemostatic containment
Host factors at high altitude? Check. Tissue hypoxia is the core inciting problem in acute mountain sickness. Pathogen factors? Check. Lipopolysaccharide has been detected in the blood of high altitude mountaineers, possibly because of increased gut permeability resulting from exertion and hypoxia. Tissue disruption/injury at high altitude? Check? Tissue factor is elevated in climbers, particularly after exhaustive exercise. These exposures drive potentially useful inflammation in wounds, helping to trap pathogens. To an immune cell, high altitude resembles an acute injury. The similar signal environment is what drives pathological inflammation and tissue edema.
The key regulator of inflammation during hypoxia is hypoxia inducible factor. Hypoxia inducible factor (HIF) is a transcription factor that acts as an oxygen sensor. The heterodimer that makes up HIF exists in all mammals, and the functional equivalent of HIF exists in all metazoans, suggesting that its oxygen sensing function is conserved across widely phylogenetically distant organisms. In other words, HIF is doing something important. Does HIF exist just to cause acute mountain sickness? Of course not. HIF is shaped by selection at low altitude to regulate metabolism and immune function. Its function is adaptive during wounds, day-to-day trauma and infection. One important adaptation to hypoxia accomplished by HIF-1 is reduced oxygen consumption by mitochondria in human cells. This in combination with a shift in cell metabolism reduces cell death in hypoxia.
My main take-away is: High altitude hypoxia resembles an infection-prone state. The immune system responds in ways that resemble what happens during an infection. Inflammation that is potentially beneficial at low altitude is maladaptive at high altitude. This misfire of the immune system is classic example of gene-environment mismatch.
When confronted by a low oxygen challenge, it might be possible to predict when a response will be adaptive, and when it will be maladaptive. Although it was my understanding that there would be no math, the likelihood of a functional adaptation is a function of the strength and duration of selection for a given condition, predicted by the following:
Duration of time that natural selection has acted on the trait expressed as number of generations
Exposure risk for an individual for a specific condition – the frequency and length of exposure – like pack years of smoking.
Fitness consequences of the event
Size of the population at risk
putting them together: D*E*F*S
For high altitude hypoxia, the Duration would be several tens of thousands of years. The Exposure risk for an individual is 100%. Fitness consequences of hypoxia include reproductive consequences (e.g. in utero growth retardation), capacity for physical exertion at altitude, and disease risk (acute mountain sickness). These are considerable. The Size of high altitude populations varies, numbering in the tens to hundreds of thousands, changing also over time. The populations are not so small as to risk stochastic disappearance and are large enough to contain genetic variation that permits adaptive genetic change.
For high altitude poplations, D*E*F*S will be high, perhaps explaining the selection that permits adaptation to hypobaric hypoxia in Andeans, Tibetans, and Ethiopians. For a sea-level population, D*E*F*S will be zero (or minimal but non-zero if accounting for occasional rare trips to altitude).
However, hypoxia is not just altitude related. Hypoxia related to lung infection, trauma, or anemia have their own D*E*F*S calculation. Further different exposures/conditions may require different adaptive responses. These compete with each other, and adaptations might come at the expense of other adaptations. Thus, Tradeoffs must enter the equation.
(DEFS)/T
Acknowledging tradeoffs is simply a way of understanding that nothing is free in biology or in medicine or in evolution. There are costs inherent to each adaptation.
For high altitude people, adaptation to hypobaric hypoxia might possibly come at the expense of adaptations to other sources of hypoxia. One potential tradeoff involves the hypoxic response in sepsis. We will explore this next.
Next up – what does mountain sickness have in common with sepsis, and what does that mean for evolution?
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
3 replies ›