If we view fat purely through the lens of pathology, we are blind to the adaptive function of fat.
It is popular to pathologize fatness, and to emphasize the burden of disease that accompanies excessive body weight. Especially when individuals are blamed for being fat, the notion that fat is a disease and a moral failing causes more, not less, human suffering. In addition to fat stigma, another issue with labeling fat as disease is that we can lose sight of why fat evolved in the first place.

What is fat for? Humans, especially human infants, are among the fattest animals at birth on the planet, and for good evolutionary reasons that Chris Kuzawa has explained. These include nutritional buffering of the brain and a benefit in surviving diseases during infancy and at the time of weaning.
At all ages, we depend on specialized fat storing cells, and humans have evolved a regulatory apparatus that interacts with our environment to control the quantity and kind of fat we store in our bodies. Fat evolved to be a energy storehouse and an insurance policy against difficult times. Where fat is stored on the body has a social signaling function and serves a role in attracting mates. Fat also functions as an endocrine organ that regulates a wide variety of body functions, including fertility, appetite, thermoregulation, metabolism, and immunity.
Immune cells are abundant in fat. Fat houses white blood cells, including macrophages and memory T cells. Yasmine Belkaid published a study in Cell showing that white adipose tissue is a storage site for memory T cells. These T cells are critical for antimicrobial defense, particularly when organisms are re-exposed to pathogens. In the Cell paper, transferring “white adipose tissue from previously infected mice was sufficient to protect uninfected mice from lethal pathogen challenge.” These and similar findings show that fat has an underappreciated immunologic role. This is the function of fat has recently been highlighted in studies of inflammatory bowel diseases, like Crohn’s disease.
Surgeons peering through an opened abdomen of patients with Crohn’s disease often see a characteristic pattern of fat that appears stuck to the intestine, often completely surrounding it. The fingerlike projections that grip inflamed sections of intestine were named “creeping fat” by the gastroenterologist Burril Crohn in 1932 who is also credited with first describing this inflammatory gut disorder. In 2020, Suzanne Devkota and colleagues published a Cell paper highlighting the microbiota as a primary reason why Crohn’s disease patients develop creeping fat.
This phenomenon can be understood as fat trapping and containing bacteria to the location where they belong – inside, not outside the intestine. In Crohn’s, bacteria are often threatening to spill out of the intestine, into surrounding organs and the bloodstream. Creeping fat works like an inner tube patch, sealing perforations in the gut that would otherwise cause bacteria to enter parts of the body that we would prefer to be sterile (or at least as sterile as possible).
“The primary role of adipose as a storage form for excess calories, while necessary, suggests a passive role in the body. However, we show here that adipose plasticity may have another, equally important, purpose that involves protecting the body from dissemination of harmful antigens at sites of inflammation or injury.” – Ha et al. Cell 2020
The underlying problem in Crohn’s disease is an immune deficiency. Affected individuals have immune deficits that make it hard or impossible to keep gut bacteria where they belong and where they are tolerated in abundance. Because of those deficits, gut bacteria escape from the intestine more readily. Although some argue that the inflamed fat is part of the problem in Crohn’s disease, a more parsimonious view is that creeping fat is a protective backup mechanism, deployed only when primary defenses fail to keep gut bacteria in check. Of course, we will often observe an association between the quantity of inflamed mesenteric fat and the severity of disease. But association is not causation. The underlying cause of creeping fat is bacteria on the move; the reason for escaping bacteria in Crohn’s disease is a problem with the immune system. At it’s essence, Crohn’s is a deficit in the body’s ability to corral and control the microbiome.
In other conditions, including appendicitis, fat serves a functional role that physically blocks the escaping bacteria. Fat appears bright on CT scans of patients with appendicitis, highlighting the functional inflammatory role it has in sealing up abscesses in the gut. Inflamed fat produces chemokines and cytokines, and becomes metabolically activated to fuel activated immune cells that mop up the bacteria that become embedded in the fat. Some fat cells, pre-adipocytes have been shown to gobble up of bacteria, a process known as phagocytosis, that find their way into fat.
“In addition to the immune cells of adipose tissue, evidence is emerging that even pre-adipocytes and adipocytes are likely to interact with invading microbes.“ – Hegde and Dhurand 2013
Even in those of us without inflammatory bowel disease, fat surrounding visceral organs is important in containing dangerous microbes in the microbiome. For this reason, unhealthy microbiomes trigger an expansion of visceral fat around the midsection. This viewpoint also explains why there is a relationship between dysbiosis, insulin resistance, and visceral fat. And it explains why probiotics have been shown to reduce the accumulation of abdominal fat. When there are fewer harmful gut microbes, there is less need for defensive fat.
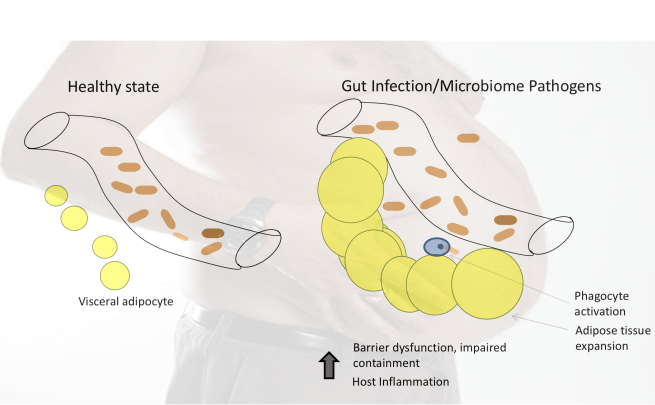
The key to understanding abdominal obesity is the immune function of fat, and its role in fighting gut infections and containing unhealthy microbiomes. Accumulating belly fat is a predictable response – not just to excess calories in the diet – but also to microbial threats. It’s immune function is a two-edged sword that can protect us, but it can also cause smoldering inflammation that has long term costs to human health.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
I really enjoy your posts on the gut microbiome and this one sort of sums up your past posts–particularly the ones on the role of fat. Great job! It forced me to study the anatomy of visceral adipose tissue in both humans and pigs. I am part of a project in the Netherlands to study the function of the cecum in pigs and this has helped in our understanding–there is very little known about the cecum and how it impacts the microbiome. Detailed information on visceral fat may be a subject in human medical school but it is not a requirement for study in animal or human nutrition; I hope more people start to understand the importance. I consider the entire body of an animal as part of immunity and this fits into that concept–I really don’t like viewing immunity as separate. Metabolism and immunity are so interrelated biochemically that is almost impossible to separate the two from each other.
We have been experimenting and implementing formulation techniques that take advantage of your concept of host-microbe conflict. I have about 3 years of data from large swine farms that [confirm] these ideas can be used to help our animals remain in better health and reduce their negative impact on the environment. Everyone, or at least almost everyone, is interested in the microbiome but I am not sure many are trying to utilize the idea of minimizing host-microbial conflict to improve its function.
Fred
Brilliant insights, Fred. I 100% agree with the idea that the entire body of an animal is part of immunity. Ed LeGrand has made a similar observation, which is that an animal should take full advantage of all of its resources (e.g. all of its body) when confronted by a life threatening challenge. Ed feel free to jump in here!
Interesting perspective on abdominal. Perhaps certain bacteria trigger this? I’m thinking of populatins exposed to plenty of microbes, thin, and no abdominal fat (obviously, at least).