I have been thinking a lot over the last decade about what features of the microbiota really matter. On the one hand, myriad studies using germ free animal models have shown powerful effects of the microbiota on physiology – with evidence for a causal role of the microbiota in obesity, hypertension, cardiovascular disease, cancer, and neurodegenerative diseases. Here we have evidence that the microbiota are involved in virtually all of the things we care about as physicians and as patients. On the other hand, human studies are wildly inconsistent. Most of the time, diseased patients do not show an overall change in the microbiota community structure or consistent differences in specific taxa compared to those without disease. For example, I just published a review paper on the role of the microbiota in obstructive sleep apnea (OSA). I will write more on this in a separate post but the bottom line is this: human studies do not agree on which, if any, alterations of the microbiota occur in OSA. Because of this, it is hard to say whether and how the microbiota is actually involved. Additionally, some of the largest human intervention studies focused on the microbiota – e.g. probiotics for diarrheal illness – have had disappointing results. We are left with a confusing situation. You’d be justified in thinking the microbiota is the one of the most important factors in diseases. Also, if your reaction is “meh, the microbiome is overhyped” you can also find some justification. I think reality lies somewhere in between.
Part of the problem is that a multitude of microbiota outcomes are reported in the literature and we don’t know which are most important. Is it alpha, beta, Shannon diversity, or functional gene changes, or the transcriptome, or the metabolome? What actually matters? From an evolutionary perspective the thing that matters most is fitness – how does the microbiota influence the fitness of its host. It is rare, but not unheard of, for a microbiota study to report fitness. Survival is a stand-in for fitness that is reported in some studies of critical illness. However, most human studies involving microbiota do not report survival, for the happy reason that, most of the time, death is uncommon.
A recent study caught my eye because it focused on the resistance of the microbiota to colonization by the pathogen Salmonella enterica serovar Typhimurium (S.Tm). S. Tm is a nasty enteropathogen that causes a diarrheal illness that is sometimes lethal. Infection by S. Tm, then, is a fairly good proxy for fitness. This study looked at the effect of non-antibiotic drugs – ranging from the cholesterol lowering drug simvastatin to the antihistamine terfenadine – on the microbiota in mice. Although it is well known that antibiotics can affect the probability of pathogen overgrowth and infection (e.g. by C. difficile), the risk from non-antibiotics has drawn little attention.
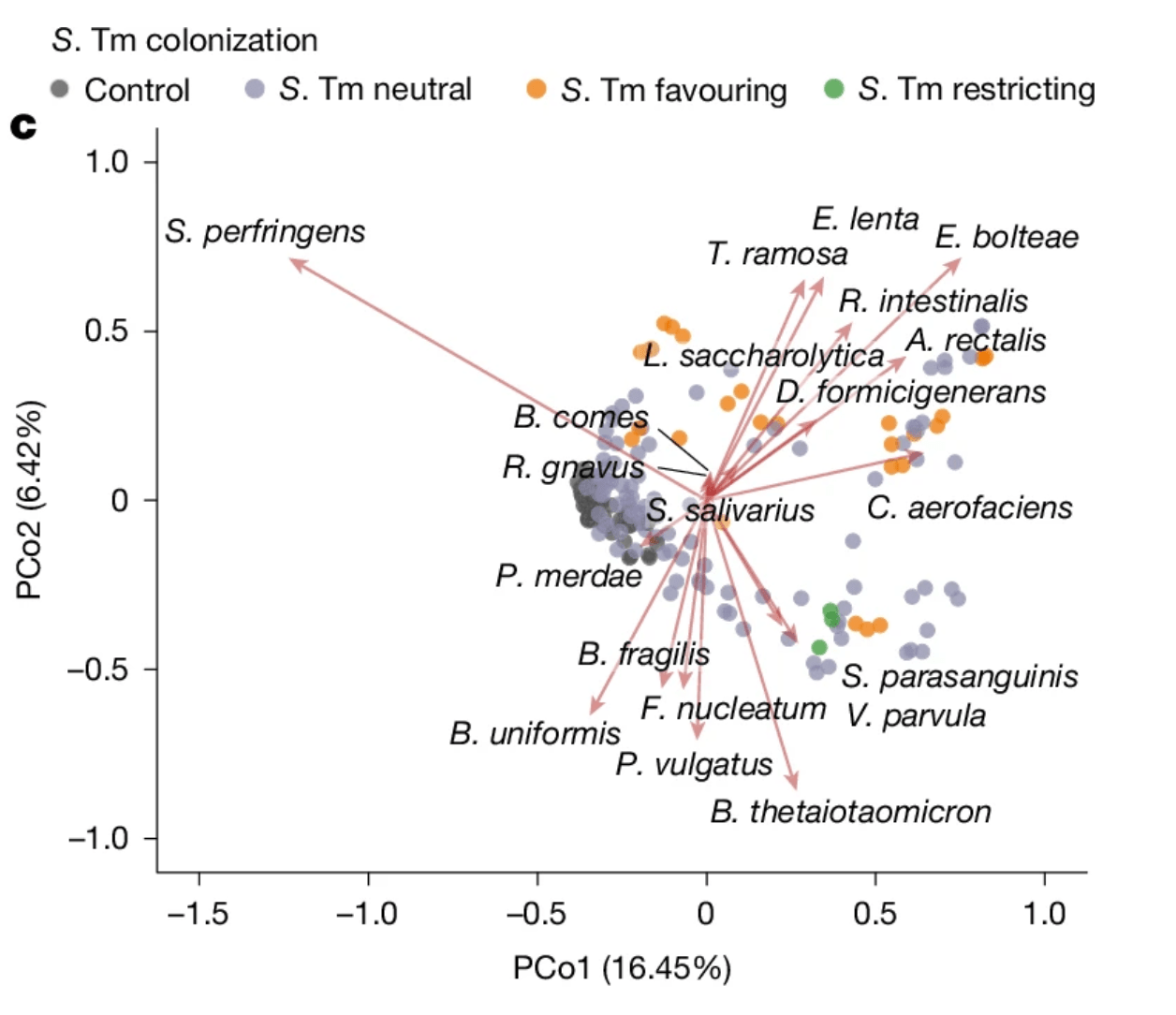
In this study by Grießhammer et al. non-antibiotics did influence the microbiota. Non-antibiotic drugs tended to disadvantage benign commensal bacteria, resulting in lower numbers, while boosting the numbers of gamma-proteobacteria. Gamma-proteobacteria is the group that contains many medically-important human pathogens – think invasive E. coli and meningitis-causing Neisseria meningitidis. S. Tm is another member of Gamma-proteobacteria. The use of non-antibiotics increased susceptibility to colonization and infection by S.Tm in this study. While this was a mouse study, it has implications for humans, especially those taking multiple medications. It is very likely that commonly taken medications – such as hormones, anti-histamines, cholesterol drugs – influence the chance of colonization and infection. This outcome is relevant to fitness because most deaths historically have resulted from infection. Colonization resistance, then is an evolutionarily relevant metric of fitness.
If this notion is correct, it might explain why some drugs have off-target effects on inflammation and metabolism. It is possible that hosts compensate for increased susceptibility to infection by increasing investments in innate immunity. Those investments come with costs, including those related to chronic low grade inflammation. I agree with Grießhammer et al. that the dangers of polypharmacy might happen because of these harmful effects on the microbiota. Microbiota researchers would be well advised to pay more attention to the effect of non-antibiotic drugs on outcomes of their studies. Microbiota-effects of non-antibiotic drugs are a largely unmeasured and unappreciated source of confounding and inter-individual variability. Finally, colonization resistance matters for the microbiota and host; this outcome deserves more respect and more attention from microbiota researchers.
References:
Alcock J. Obstructive sleep apnea and the microbiota, mechanisms and opportunities. Current Opinion in Pulmonary Medicine. September 2025. DOI: 10.1097/MCP.0000000000001211
Grießhammer, A., de la Cuesta-Zuluaga, J., Müller, P. et al. Non-antibiotics disrupt colonization resistance against enteropathogens. Nature 644, 497–505 (2025). https://doi.org/10.1038/s41586-025-09217-2
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
Leave a comment