The two main evolutionary concepts I teach students about aging are George C. Williams’ idea of antagonistic pleiotropy and the declining power of selection hypothesis (aka selection shadow hypothesis) proposed by J.S. Haldane and Peter Medawar in the 1940s and 1950s. Medawar posited that beneficial genes, coding for body maintenance and repair, are selected for more strongly at early ages (pre-reproduction) than after reproductive age.
For this: imagine a hypothetical gene that prevents cancer at age 10 and another gene that prevents cancer at age 100. The gene preventing cancer after age 100 will not have any effect on most people for the simple reason that most people are dead before reaching 100. This is still true even if you take senescence out of the equation; random accidents will claim many lives. More people survive to age 10 than age 100, so a gene that prevents cancer at young age will be expressed and selected for. The gene expressed only in extreme old age will be invisible to natural selection.
Medawar extended this idea to include mutation accumulation. This idea posits that the body accumulates deleterious mutations that take effect at older ages. Because of the declining power of selection, these mutations are not selected against. In wild populations, not enough organisms reach advanced age, so these mutations are invisible – e.g. they fall in a selection shadow. If an individual does achieve advanced chronological age, these mutations exert damaging effects, reducing fitness and contributing to senescence.
When I bring up mutation accumulation with students, I ask them to consider imaginary genes with mutations that have deleterious effects at different ages. A harmful mutation that shows up at an impossible age, say 150 years old, would never be subjected to negative selection because no human has ever lived to that advanced age. A mutation that has harmful effects in more realistic old age (70s-80s) would see some selection, but vastly less compared to mutations with harmful effects during youth.
We no longer need to speculate about age-dependent harmful genes. A recent paper by Liu et al., showed they exist! The genes that were studied in this paper were not technically human at all. They originated in viruses that inserted their genes into our genomes. These endogenous retroviruses introduce genes that Liu et al showed cause cellular and tissue senescence at older age.
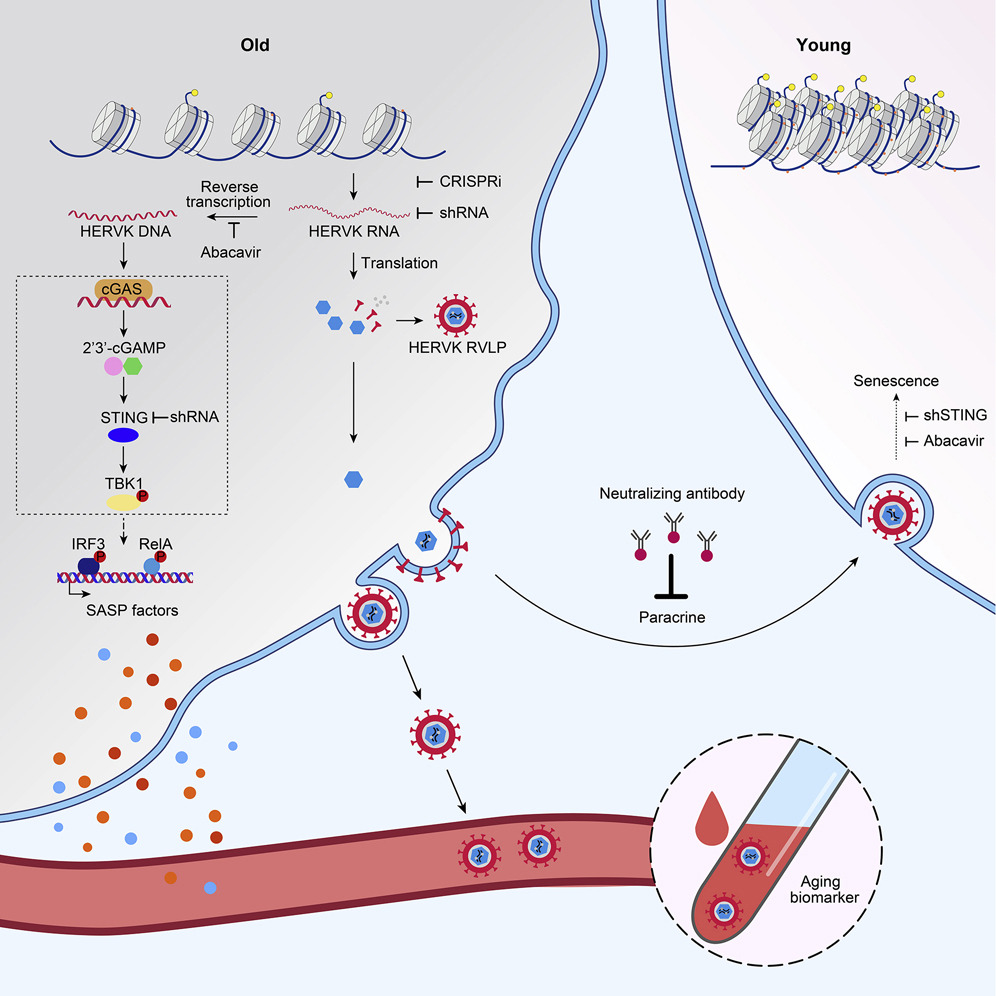
From the abstract: “Whether and how certain transposable elements with viral origins, such as endogenous retroviruses (ERVs) dormant in our genomes, can become awakened and contribute to the aging process is largely unknown. In human senescent cells, we found that HERVK (HML-2), the most recently integrated human ERVs, are unlocked to transcribe viral genes and produce retrovirus-like particles (RVLPs). These HERVK RVLPs constitute a transmissible message to elicit senescence phenotypes in young cells, which can be blocked by neutralizing antibodies. The activation of ERVs was also observed in organs of aged primates and mice….These findings indicate that the resurrection of ERVs is a hallmark and driving force of cellular senescence and tissue aging.”
Late life awakening of genes with a negative effect on survival are equivalent to Medawar’s age-dependent deleterious genes. We do have age-dependent deleterious genes, but in this case they are not even human; they come from viral elements inserted into our genomes.
In younger individuals, the negative fitness consequences of endogenous viral genes are handled by the immune system with neutralizing antibodies – but not so much in older individuals. Effectively, these harmful genes are expressed at older age but not at younger ages, because of the declining power of selection with age.
This also illustrates genomic conflict responsible for senescence. We can add aging to the list of problems that generated by genetic conflict, along with disorders of pregnancy, cancer, chronic diseases related to the microbiome, and infectious disease.
Endogenous retroviruses can be used as a biomarker of aging. Levels of endogenous retroviral elements were higher in the serum of old people. Liu et al. also found higher retroviral activation in joint cartilage where it might have an effect on osteoarthritis, a common joint pathology of aging. Even more interesting, they found that repression of endogenous retrovirus can attenuate aging! Adding the drug Abacavir in the drinking water of mice increased grip strength, improved physical condition and improved memory in mice. Abacavir is an inhibitor of retroviral transcription. Perhaps a similar strategy can delay aging in humans. Leave your thoughts in the comments.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
EMPH Published an article by Steve Austad on antagonistic pleiotropy
Austad SN, Hoffman JM. Is antagonistic pleiotropy ubiquitous in aging biology? Evol Med Public Health. 2018 Oct 11;2018(1):287-294. doi: 10.1093/emph/eoy033. PMID: 30524730; PMCID: PMC6276058.
How to measure grip strenght in mice?
Thanks for the link on antagonistic pleiotropy by Austad and Hoffman. It is a good one to share with students. As for grip strength in mice that is a good question!
I found the grip strength information from the paper, something called PanLab grip strength meter: “grip strength of the mice was assessed using a Grip Strength Meter (Panlab Grid Strength Meter, LE902). Briefly, four limbs of the mouse were placed on the top of the Grip Strength Meter and pulled along the direction of the grid at a constant rate until the Grip Strength Meter was released by the mouse. This process was repeated 10 times and the peak pull force at each time was recorded on a digital force transducer. The mean values from 10 consecutive trials were taken as the grip strength of each mouse.”