Updated!
The promise and power of science is that it produces results better than superstition, anecdote, and intuition. Vaccines, antibiotics, and insulin are powerful examples of those results. However, success can sometimes lead to the phenomenon of overshoot. Overshoot is when treatments miss the mark, and cause more harm than good. One example of overshoot is when drugs and therapies work for a narrowly tailored population but then are misused when extended more broadly to groups for which they were not intended. Overshoot can happen when physicians see pathology where it may not exist. Because not all the changes that happen in a disease are pathological, this can lead to overtreatment. Overshoot also applies to the failed paradigm of research that is myopically focused on the molecular mechanisms of disease. This often leads to therapies that don’t work. Too often we are encouraged by pre-clinical and early stage trial results that are followed by large scale randomized controlled trials showing no effect or patient harm. Not infrequently, a single successful trial later fails to be replicated. Replication failure has resulted in the withdrawal of a drug from the market. Overshoot results in overtreatment and overdiagnosis, major sources of wasteful medical practices and patient harm. According to a BMJ assessment of 3000 randomized clinical trials, only 11% of common medical practices showed evidence of benefit, 24 % were deemed to be likely beneficial. The majority was either harmful or had no evidence to support it.
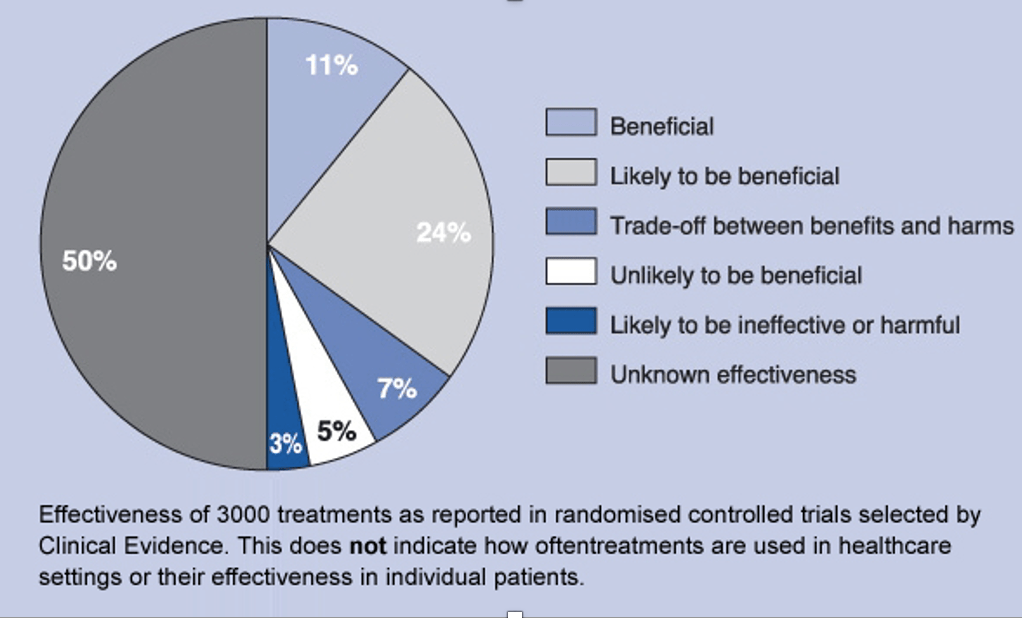
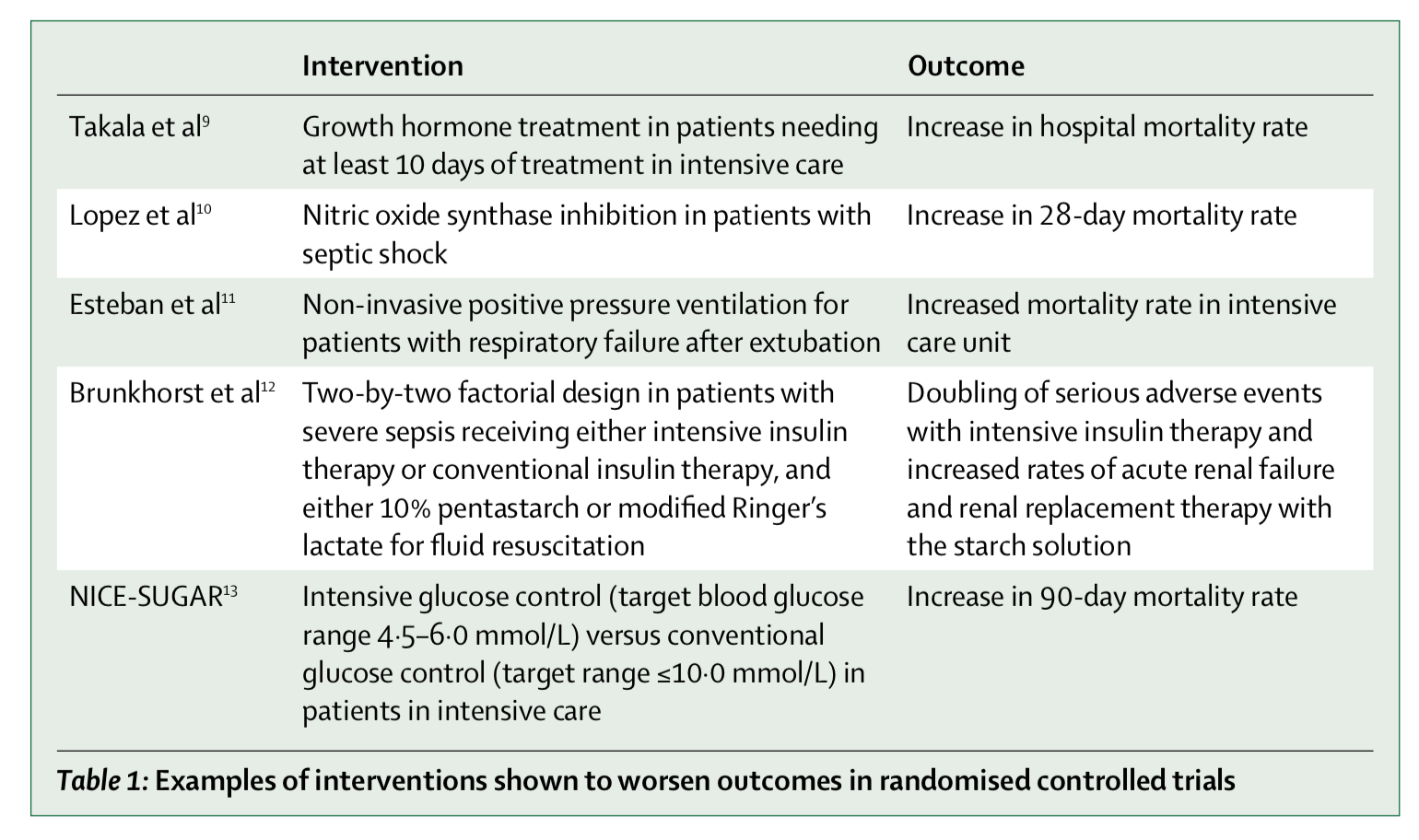
In line with these findings, the authors Vincent and Creteur pointed out that a great many advances in in critical care medicine have been accomplished by monitoring less, intervening less aggressively, and avoiding iatrogenic harm to patients in the intensive care unit. I wrote about the pitfalls of excessive monitoring of patients in the previous post on this site. We should be doing more with less, Vincent and Creteur argue. In the 8 years since that article was published, even more evidence has emerged that once-promising strategies for critically ill patients can do more harm than good. Often, evidence that is contrary to preconceived notions is dismissed or overlooked. See for instance the FEAST trial by Kathryn Maitland, in which critically ill children in three Sub-Saharan African countries were randomized to standard intravenous fluid resuscitation versus usual care without fluid resuscitation. In that study, providing U.S./European standard of care with fluid resuscitation resulted in increased mortality, a result that surprised even the investigators. Unfortunately this provocative result did not lead to further trials of the effectiveness of intravenous fluid resuscitation in children in industrialized countries. Worse, the World Health Organization initially ignored her findings. “WHO remained silent and only later modified its guidelines, and even then not adequately” according to Maitland.
When faced with the uneven progress of biomedicine, it might be time to return to first principles. By first principles, I mean evolutionary principles. Using evolutionary reasoning, when can we expect a drug or therapy to work? Conversely, when should we be most doubtful about an intervention?
Consider first: there are good reasons to think that we can intervene and improve mortality during infection. One reason is that pathogens evolve. As vividly exemplified by the ever-evolving and immunity-evading SARS-CoV2 variants, our immune toolbox is never perfect. This means that we will never see an organism perfectly adapted to resist lethal infection. There are other important reasons for imperfect adaptation to infection. These include energetic or biological constraints, aging, prior injury, chronic infection, other environmental challenges, such as the mismatch between the modern and ancestral environment. (One important mismatch that is rarely considered is the fact that the intensive care unit and its interventions are a novel environment for which we are not evolved.)
Despite the ongoing evolution of host defenses, we can infer from the constraints and mismatches discussed above that our immune systems are always imperfect. Any patient trait (for example, C-reactive protein, neutrophil count, blood pressure, body temperature, and so on) is probably not precisely at its theoretical optimum. For an enterprising interventionist this would seem to be great news. Imperfect patient parameters would present a nearly infinite number of potential targets for intervention. How exciting!
Now we should consider why these juicy targets might not be so great after all. John Marshall wrote eloquently about the myriad targets for intervention in sepsis that proved to be dead ends. More recently, I suggested that the definition of sepsis as a dysregulated state is not substantiated by evidence and needs re-appraisal. The main reason why intervening on the immune system itself may not work as intended is the cumulative effect of natural selection acting on the immune system and its regulation. This produced a functional result that may be better than we’d expect from looking at any of its individual parts. Several evolved features make the body resistant to improvement. First, our defenses co-evolved with evasive and manipulative pathogens; our bodies anticipate manipulation and the immune system has evolved ways to compensate if any one part fails. We observe a great deal of immune redundancy, a capacity to correct and compensate, and parallel regulation of defensive systems. Our imperfect host defenses work seem to function “well enough” much of the time. (Though not always, of course).
For all these reasons, many of the the drugs we have high hopes for in the laboratory might not work as well in real patients in the real world.
For any drug aimed at the immune system to work, the target and the drug need to satisfy the following conditions:
1) The trait must be non-optimal and we must know the direction in which the trait needs to be adjusted, in an individual and on average in a population.
2) Our therapy must be superior to the body’s own capacity to regulate the trait. (In other words, the trait must actually be dysregulated or maladaptive instead of the alternative, that it is regulated and adaptive).
3) Changes in other physiologic systems have not adequately compensated for the trait.
4) We can avoid unintended consequences. By “correcting” the trait, we do not undermine other host compensations or make other traits suboptimal.
5) The intervention will have a predictable and beneficial effect despite individual trait variation.
6) Pathogens will not benefit from the trait change caused by our therapy.
7) Pathogens will not evolve to exploit the trait change or intervention.
Satisfying these assumptions is a heavy lift. That’s why no magic-bullet immune drug has worked to date for critically ill septic patients in the intensive care unit.
On the other hand, evolution can inform when we can expect therapies to work. Applying evolutionary reasoning may lead us to a possible effective therapies:
1) We can remedy a constraint, e.g. provide supplemental oxygen for severe hypoxia, provide glucose during hypoglycemia, and reverse dehydration from diarrhea.
2) Target virulence and manipulation by pathogens that subvert the immune system and block our defenses.
3) Manage trade-offs involving variation in immune genes and the microbiome, for instance.
4) Recognize deficiencies related to senescence.
5) Minimize gene-environment mismatch, specifically we have not evolved to be in hospitals or to undergo intensive care unit level of care. We can ameliorate this mismatch by not interfering with sleep or circadian rhythm, and by minimizing stress, excessive light, and noise (think alarming monitors, etc) that disturb our patients.
6) Lastly and most importantly, research efforts in infectious disease are best targeted at the infecting organism, not the host. This is accomplished most effectively with vaccines that provide durable protection, and also from antibiotics and antivirals. Each of these comes with the unavoidable tradeoff and consequence of resistance evolution, because pathogens evolve faster that we do, and often faster than we can develop new antibiotics and vaccines.
Designing new treatments, based on evolutionary first principles, is only half of the solution to overshoot. The other half is identifying existing treatments that lack evidence and potentially are harmful. Evolutionary first principles can highlight drugs and treatment strategies that are unlikely to work or are paradoxical when considered from an evolutionary lens. Those can be flagged for further scrutiny, and useless treatments then discarded. In that way, evolutionary first principles can be a key way to achieve better health care by doing less of it.
Copyright © Joe Alcock MD
Key references:
Epstein D. When Evidence Says No But Doctors Say Yes – The Atlantic 2017
Maitland et al Mortality after Fluid Bolus in African Children with Severe Infection N Engl J Med 2011; 364:2483-2495
Marshall. Why have clinical trials in sepsis failed. Trends Mol Med 2014 Apr;20(4):195-203.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
Leave a comment