Sepsis is an important cause of mortality, causing an estimated 60,000 deaths yearly. Sepsis is also expensive to treat, and is associated with expensive medical procedures, such as life support and intensive care. Despite advances in supportive care over the last 30 years, the mortality rates have remained stubbornly high, as much as 30-40%.
In septic shock, treatment includes early and appropriate empiric antibiotics, source control, and supportive care. Of these, antibiotics and source control are thought to be the most important, but surprisingly little data support the elements of care. Controversy surrounds most aspects: fluids, pressors, oxygen replacement, and enhancement of cardiac output, to name a few. Kox et al wrote an illuminating recent review on aggressive therapy in sepsis and critical illness entitled Less is more in critically ill patients. In light of these ongoing controversies, can an evolutionary perspective of disease provide any guidance?
One example involves fluid administration:
“Recent research in sepsis has also hinted at a phenomenon thus far not even considered—that hypotension is an adaptive response, honed over millions of years of evolution, which may mean that we are actually making patients worse when we thought we were helping them.“-David Anderson
Read Anderson’s account of controversy in giving fluid therapy for sepsis and critical illness.
And: Conservative Fluid Management In Intensive Care Medicine by David Gattas
A second, related possibility is that the immune systems response is also adaptive in septic shock. If so, anti-inflammatory treatments won’t work. Supporting that notion is the recent clinical experience with recombinant activated protein C:
Persistently high sepsis mortality prompted many researchers and funding organizations to seek anti-inflammatory treatment to decrease the apparently harmful immune effects of sepsis, which has been described as an out of control inflammatory response.
One thing that biomedical researchers noticed was that septic patients who die often had low levels of activated protein C. A recombinant form of activated protein C, called Xigris, has anti-inflammatory and anticoagulant properties. Since inflammation was thought to be out of control in the systemic inflammatory immune response, it stood to reason that an inhibitor of inflammation and clotting might reduce deaths in sepsis.
In 2001 the PROWESS study appeared to show just that.
10 years late, on October 25, 2011 the FDA recommended that Xigris be withdrawn from the market. Remarkably, the company selling Xigris, Eli Lilly, was able to profit from this ineffective drug for nearly the entirety of the time Xigris was under patent. The long road to arrive at this conclusion is an interesting story:
Epitaph for Xigris – I never worked a day in my life!
What lesson from Xigris’s failure can we learn about the functioning of the immune system, about sepsis, and evolution? Notably, Xigris is not the only high-profile failure for sepsis immunomodulatory therapy. A more recent anti-inflammatory therapy, a toll-like receptor TLR-4 blocker known as Eritoran, also failed spectacularly in clinical trials.
Not be deterred, a new therapy for sepsis targeting the pro-inflammatory cytokine TNF-alpha, based on a compound called CytoFab, was recently tested. How do you suppose that one went? Shockingly, it too failed.
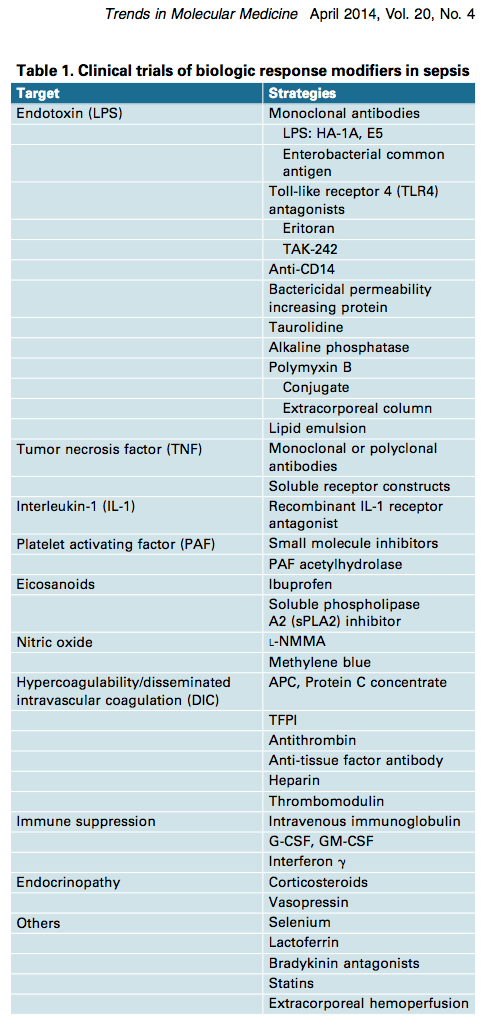
Table 1 showing failed clinical trials of immune and other therapies in sepsis above, and the abstract below, is reproduced from Marshall Why have clinical trials in sepsis failed? Trends Mol Med 2014
“The systemic inflammatory response is biologically complex, redundant, and activated by both infectious and noninfectious triggers. Its manipulation can cause both benefit and harm. More than 100 randomized clinical trials have tested the hypothesis that modulating the septic response to infection can improve survival. With one short-lived exception, none of these has resulted in new treatments. The current challenge for sepsis research lies in a failure of concept and reluctance to abandon a demonstrably ineffectual research model. Future success will necessitate large studies of clinical and biochemical epidemiology to understand the course of illness, better integration of basic and clinical science, and the creation of stratification systems to target treatment towards those who are most likely to benefit.”
These examples suggest that continuing to search for elusive so-called “magic bullets” in sepsis is a losing strategy. As cited in a recent article about the failure of anti-TNF-α in sepsis: “Success is the ability to go from one failure to another with no loss of enthusiasm,” a quotation attributed to Sir Winston Churchill. On the other hand, repeating same thing while expecting different results is also…well you know the cliché.
Student writing assignment:
You have a relative living in Amsterdam who was approached by a Dutch researcher about enrolling in a real study of an anti-inflammatory agent in endotoxemia: The study, entitled in vivo effects of C1-esterase inhibitor on the innate immune response during human endotoxemia is underway. What do you advise her to do and why?
Late Update: Another area of active investigation is the possible beneficial effect of the lipid-lowering drugs known as statins on sepsis and its complications. The idea is that in addition to reducing cholesterol, statins have anti-inflammatory and anti-coagulant effects. (Sounds a lot like Xigris, doesn’t it.)
The blog Life in the Fast Lane has published a nice summary of what is currently known about statins in sepsis that is absolutely worth a read. Germane to our discussion, NEJM published a recent test of statins in the sepsis-associated lung disease. Result: Failed. Last year, a randomized trial of atorvostatin in sepsis failed to show an improvement in 90 day mortality. The authors of that study hold out hope that statins will be proven efficacious in sepsis, and the final word on this topic has not been published. Another randomized trial of statins in sepsis is currently recruiting patients, what do you predict will be the results?
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine

Metabolic Theory of Septic Shock
http://www.wjgnet.com/2220-3141/full/v3/i2/45.htm