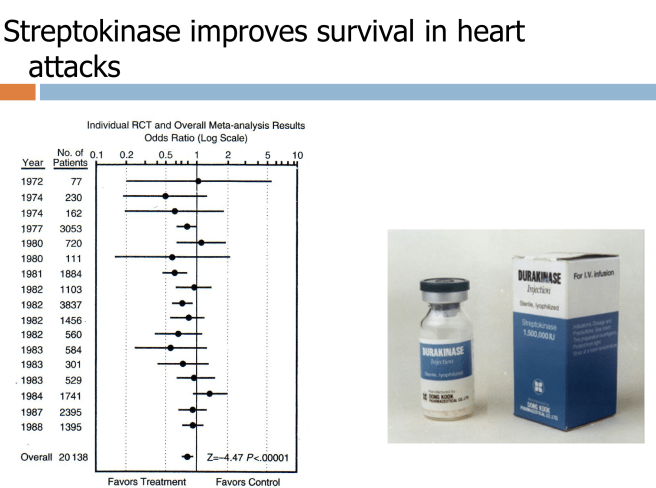
Drugs derived from microorganisms have a long history of use in medicine. One example is Streptokinase, derived from a strain of Streptococcus sp., which was found to be useful in treating patients with acute myocardial infarction (AMI). The clot busting drug Streptokinase is essentially a bacterially derived enzyme that converts plasminogen to plasmin. A series of large clinical trials in the 1970s and 1980s demonstrated a survival benefit in treating patients with acute myocardial infarction (see figure above). In AMI, streptokinase dissolves the clots that deprive the heart muscle of blood flow and oxygen. More recently streptokinase has been supplanted by a recombinant product tPA, tissue plasminogen activator, and most physicians have forgotten that this class of clot busting drugs was originally derived from bacteria.
Why do bacteria produce enzymes that dissolve clots?
A wide variety of pathogenic bacteria use the plasminogen system to invade tissues.
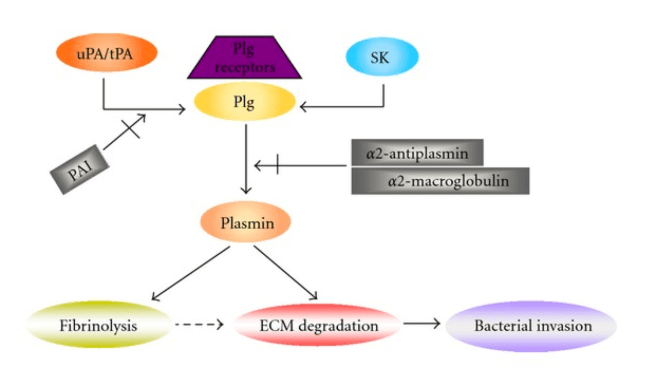
From Bhattacharya et al. Bacterial plasminogen receptors utilize host plasminogen system for effeective invasion and dissemination. J Biomed Biotech (2012)
Fibrinolysis and clot degradation are an important strategy that some bacteria use to disseminate within the host, as illustrated by the figure by Bhattacharya above. A recent article demonstrated the importance of the anti-clot virulence factors in the human pathogen Enterohemorrhagic E. coli (EHEC). Kuo and colleagues have shown that EHEC produces a serine protease EspP that degrades clot strength and promotes fibrinolysis. The title of their March 2016 paper says it all:
When human blood was incubated with EspP, it resulted in reduced activity of human clotting Factor VIII and in prolongation of protime (PT) and INR. The reduction in Factor VIII activity was due to proteolytic cleavage by EspP. At the cellular level, the EspP promotes the uptake of EHEC bacteria directly into intestinal epithelial cells, as was elegantly shown by In et al. in their 2013 paper:
Taken together these two papers show that the anti-clotting function of EspP allows bacteria to circumvent the host defense function of fibrin deposition and blood clotting, resulting in the direct invasion of intestinal cells by pathogenic EHEC. In clinical medicine, this serine protease is responsible for the bloody diarrhea seen in EHEC infection.
It stands to reason that since pathogens use anti-clotting virulence factors to estabish infection, that the host might engage in an arms race with bacteria to reinforce blood clotting when potentially harmful bacteria are present.
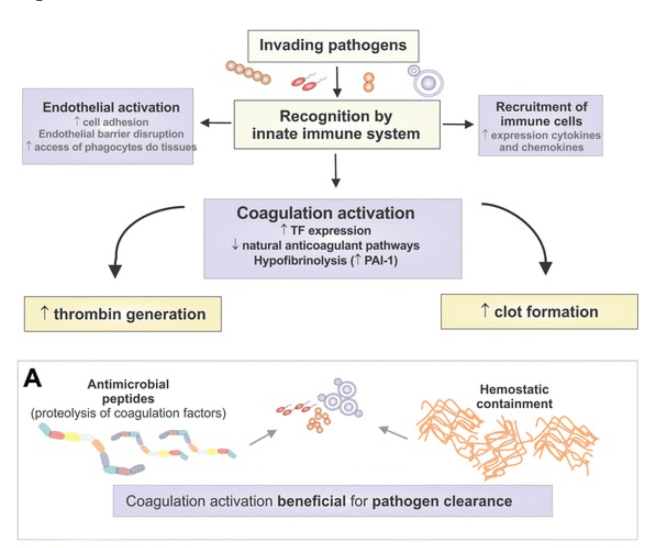
Fiusa et al (2013) wrote an elegant review in which he outlined the role of blood clotting in “hemostatic containment,” a term coined by myself and Andrew Brainard to describe the host function of blood clotting in preventing systemic spread of infection. From Fiusa et al. 2013 Causes and consequences of coagulation activation in sepsis: an evolutionary medicine perspective:
Unfortunately the arms race can go awry, with collateral damage that can damage, and sometimes kill the host during sepsis (Fiusa et al 2013) and in heart attacks and other diseases (Alcock and Brainard 2008).
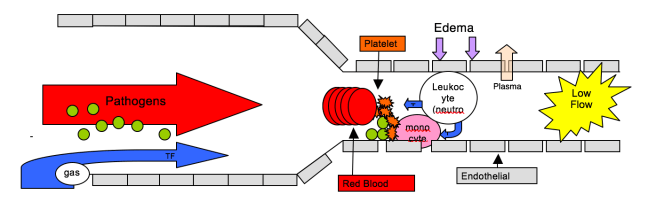
Alcock and Brainard postulated that signals from bacteria and gas that enter blood vessels following injury are potent activators of coagulation. The resulting hemostatic containment prevents bacteria from entering the systemic circulation (usually).
Since microbial signals are central to regulating the coagulation defenses of the host, we have a potential evolutionary explanation for the propensity of a variety of antibiotics to affect coagulation parameters, e.g. INR. By affecting the composition of bacteria in the gut, and at sites of infection, antibiotics may reduce bacterial signals that regulate the intensity of host blood clotting. This becomes a problem for patients taking the blood thinning medication warfarin, when a new prescription of antibiotics can sometimes lead to dangerous bleeding.
To sum up, bacteria and other microbes impair host clotting as a means of establishing infection. Host coagulation, on the other hand, is regulated by bacterial signals, and has a host defense function that is important in preventing pathogen invasion of tissues. Although we physicians have harnessed the anti-clot activity of bacterial enzymes (e.g. Streptokinase) to help patients, we should not falsely conclude that clot inhibition by bacteria is a good thing generally, or that thinning the blood during infection is cost-free (See the previous posts on Xigris here and here). Finally, we now recognize that the human microbiome influences multiple domains of host physiology, including blood clotting. What this means for human health and medicine will be left for a future post.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
3 replies ›