This is the EvolutionMedicine ‘cast #2, for July 4, 2016. This is a story worth telling.
Sepsis is an important cause of mortality, causing an estimated 60,000 deaths yearly. Sepsis is also expensive to treat, and is associated with expensive medical procedures, such as life support and intensive care. Despite advances in supportive care over the last 30 years, the mortality rates have remained stubbornly high, as much as 30-40%.
 Persistently high sepsis mortality prompted many researchers and funding organizations to seek anti-inflammatory treatment to decrease the apparently harmful immune effects of sepsis, which has been described as an out of control inflammatory response.
Persistently high sepsis mortality prompted many researchers and funding organizations to seek anti-inflammatory treatment to decrease the apparently harmful immune effects of sepsis, which has been described as an out of control inflammatory response.
One thing that biomedical researchers noticed was that septic patients who die often had low levels of activated protein C. A recombinant form of activated protein C, called Xigris, has anti-inflammatory and anticoagulant properties. Since inflammation was thought to be out of control in the systemic inflammatory immune response, it stood to reason that an inhibitor of inflammation and clotting might reduce deaths in sepsis.
In 2001 the PROWESS study appeared to show just that.
10 years late, on October 25, 2011 the FDA recommended that Xigris be withdrawn from the market. Remarkably, the company selling Xigris, Eli Lilly, was able to profit from this ineffective drug for nearly the entirety of the time Xigris was under patent. The long road to arrive at this conclusion is an interesting story.
What if the immune and coagulation responses are adaptive in septic shock? If so, anti-clotting/inflammation treatments, like Xigris, will fail. Supporting that notion is the recent experimental experience with recombinant activated protein C:
Epitaph for Xigris – I never worked a day in my life!
What lesson from Xigris’s failure can we learn about the functioning of the immune system, about sepsis, and evolution? Notably, Xigris is not the only high-profile failure for sepsis immunomodulatory therapy. A more recent anti-inflammatory therapy, a toll-like receptor TLR-4 blocker known as Eritoran, also failed spectacularly in clinical trials.
Not be deterred, a new therapy for sepsis targeting the pro-inflammatory cytokine TNF-alpha, based on a compound called CytoFab, was recently tested. How do you suppose that one went? Shockingly, it too failed.
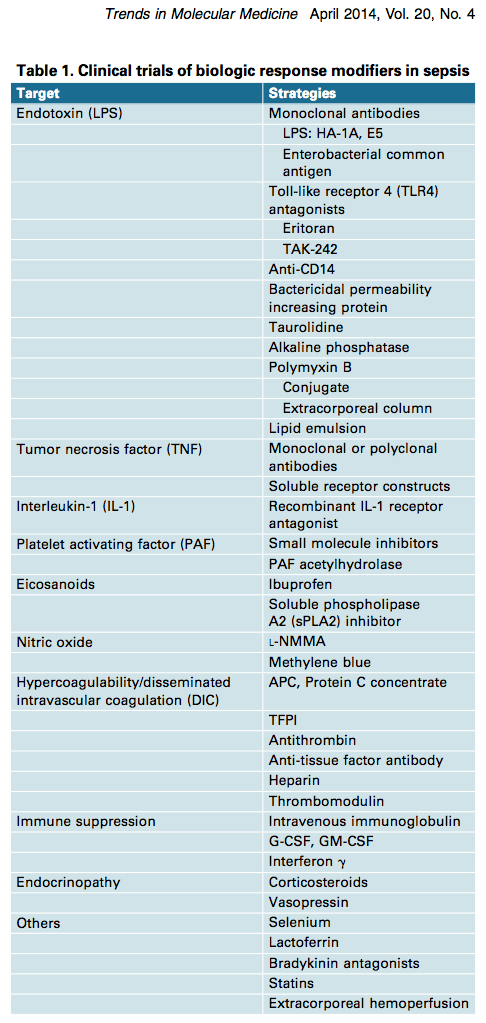
Table 1 showing failed clinical trials of immune and other therapies in sepsis above, and the abstract below, is reproduced from Marshall Why have clinical trials in sepsis failed? Trends Mol Med 2014
“The systemic inflammatory response is biologically complex, redundant, and activated by both infectious and noninfectious triggers. Its manipulation can cause both benefit and harm. More than 100 randomized clinical trials have tested the hypothesis that modulating the septic response to infection can improve survival. With one short-lived exception, none of these has resulted in new treatments. The current challenge for sepsis research lies in a failure of concept and reluctance to abandon a demonstrably ineffectual research model. Future success will necessitate large studies of clinical and biochemical epidemiology to understand the course of illness, better integration of basic and clinical science, and the creation of stratification systems to target treatment towards those who are most likely to benefit.”
These examples suggest that continuing to search for elusive so-called “magic bullets” in sepsis is a losing strategy. As cited in a recent article about the failure of anti-TNF-α in sepsis: “Success is the ability to go from one failure to another with no loss of enthusiasm,” a quotation attributed to Sir Winston Churchill. On the other hand, repeating same thing while expecting different results is also…well you know the cliché. Japanese investigators are at it again with a medication, ART-123, that works along similar lines at Xigris, at an earlier stage in the pathway leading to activated protein C. I predict the same fate for ART-123 as Xigris.
Hemostatic containment article is here
My previous post on why blood clotting is host defense and why bacteria dissolve clots
10 evolutionary mistakes physicians make
My post on underlying assumptions of sepsis treatment
Additional references from the podcast are in this file: Xigris epitaph
© Joe Alcock MD
Categories: podcasts
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
5 replies ›