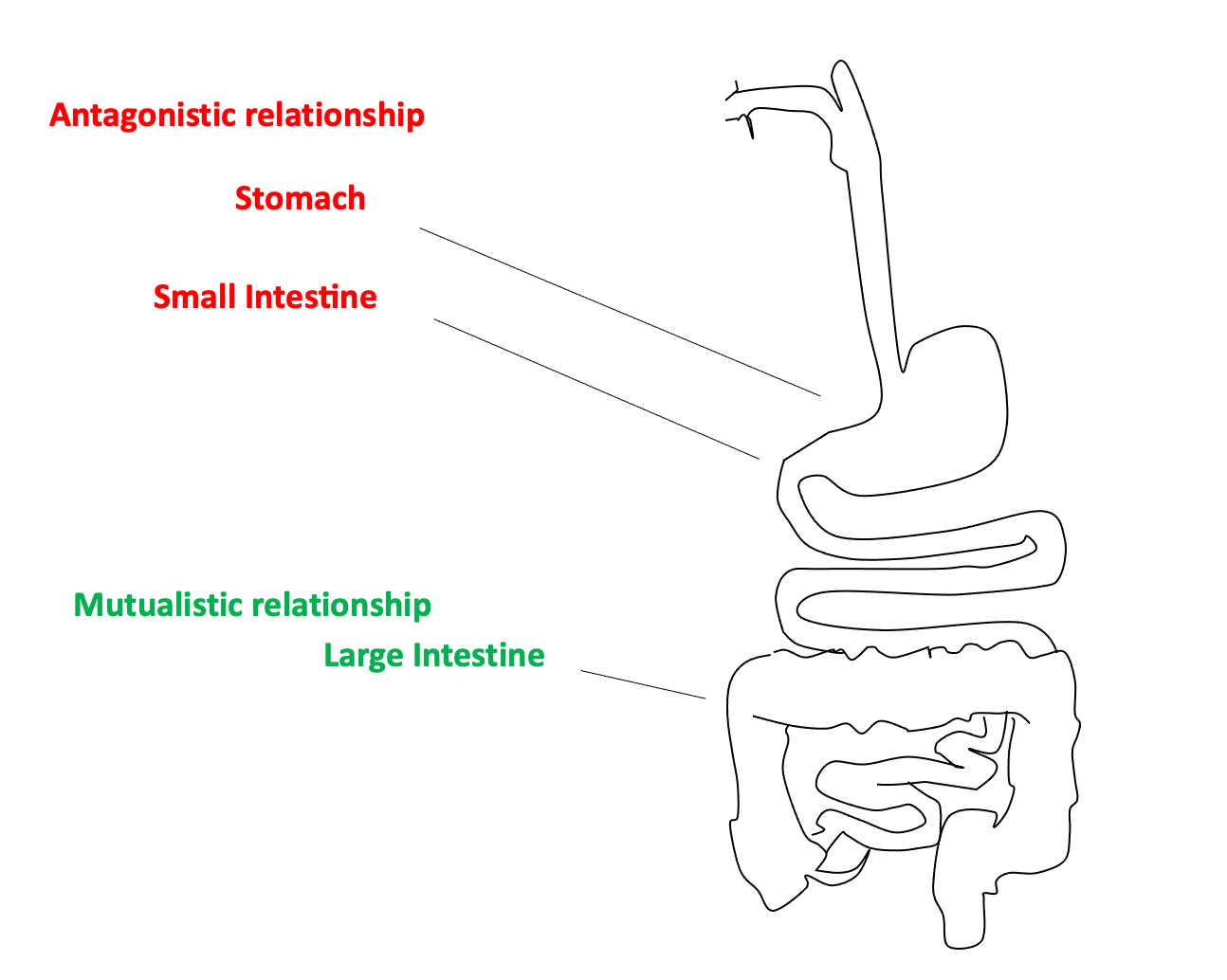
There is conflict brewing in your guts. This conflict plays out between your genome and the multiplicity of microbial genomes in the microbiota. As Herald Brüssow and others have pointed out, the conflict zone is mostly in your upper gut, above your colon. The large intestine, or colon, is relatively unconflicted, and much larger numbers of microbes are permitted by the host (host means us in this example) to take up residence there.
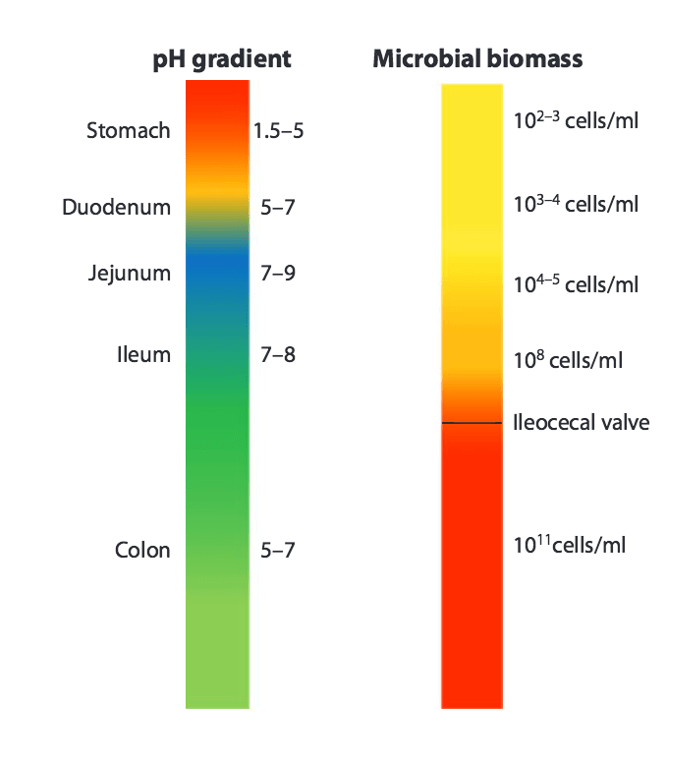
In his 2015 paper, “Microbiota and the human nature: know thyself“, Herald Brüssow wrote: “human physiology tells us that microbes in the upper part of the intestine are seen as enemies or at least undesired competitors for food, and they need to be kept at low level.” This inhibiton of microbial growth is enforced by the extremely low pH of the stomach and proximal small intestine, the production of secretory immunoglobulins in the gut, and anti-microbial bile acids.
The implications of proximal gut conflict was described also by Gaskins Collins and Anderson (2002). They describe how pathogens and their competitors have a large impact on the growth characteristics of pigs. Gaskins Collins and Anderson (2002) write: “It is curious that a class of organisms that appear to depress growth, namely Gram-positive facultative anaerobes including strains of Lactobacillus and Enterococcus, are also often used as probiotic organisms for enhancing health and promoting growth in livestock (reviewed in Jonsson and Conway, 1992). The growth-promoting effect of probiotics in livestock is less consistent than that observed with antibiotic supplementation (Jonsson and Conway, 1992). Supplementation of animals and humans with certain probiotic bacteria has been shown to provide protection against intestinal, diarrhea-producing pathogens (reviewed in McCracken and Gaskins, 2000). Therefore, probiotics may promote growth under situations in which certain pathogens are present; however, these same organisms in a cleaner facility may suppress growth via the mechanisms discussed above.”
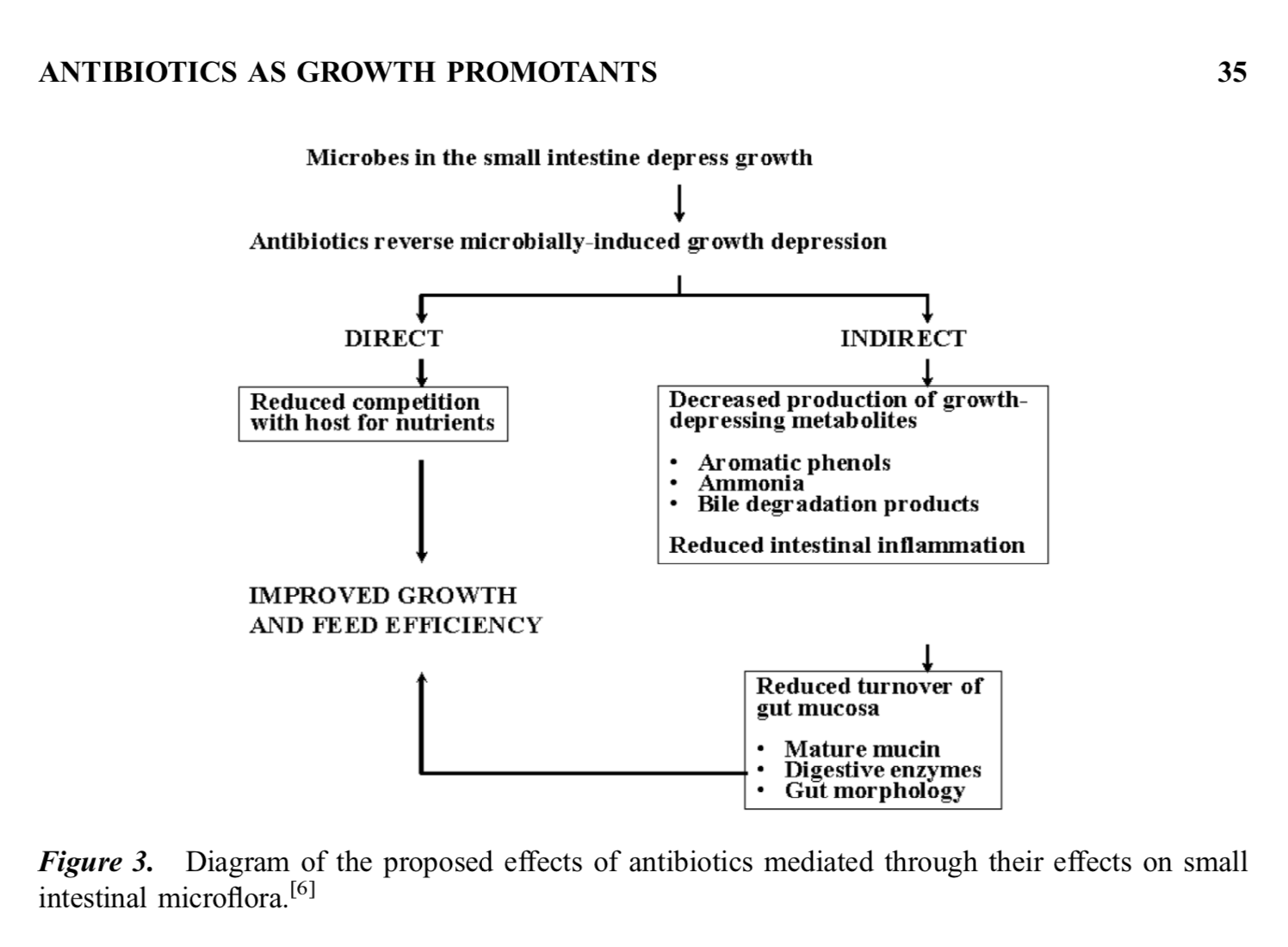
In other words, we can often get better growth performance by killing microbes, rather than adding to them. On balance, the microbiota competes with us for food. This is true even for microbes we think of as being friendly, probiotic gut microbes. Brüssow and Gastkins et al. suggest that we have a very long history of conflict with our microbiotas, one that is etched in the human gut. Similar conflict exists in the guts of many agricultural animals, including our porcine friends with whom we last shared a common ancestor 80 million years ago. This means that when things go wrong in our relationship with our microbial companions it can be for reasons both new and very old.
In future posts, we will extend this cooperations and conflict perspective to a variety of otherwise hard to explain phenomena. For instance: why is it so harmful to be on medications that raise our stomach lining’s pH? Although it is rude to point fingers, my finger is pointed directly at proton pump inhibitors here. Do we get more conflict in the microbiome overall when we remove the mutualism part, the colon, in patients who have had colectomies? Possibly. Abundance of anaerobes decreased after colon resection and the abundance of pathogens increased in one study. Does this perspective mean that animals that have a very small guts also are more in conflict with their microbiomes? Potentially, yes. Bats and flying birds have a diminished microbiome, because it is less useful to depend on microbial fermentation for energy. This is a clear cut recipe for conflict. When we humans consume foods that do not require fermentation do we run into conflict as well? Absolutely. Fiber-free industrially ultra-processed diets increase conflict, as we have written about previously, and as we might expect.
Copyright © Joe Alcock MD
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
Leave a comment