
Bifidobacterium sp.
This entry builds on the last post, which criticized overzealous antibiotic use. I argued that antibiotics are more harmful than good in self-limiting infections, and gave an evidence-based rationale for withholding antibiotics. Many of the arguments against antibiotics relate to negative effects on the microbiota.
If killing part of our microbiota is problematic, what about doing the opposite? According to the World Health Organization, probiotics are “live microorganisms which, when administered in adequate amounts, confer a health benefit.” Should we be using probiotics for hospitalized patients and outpatients? If so which ones and when?
From an evolutionary standpoint, there is good reason to expect benefit from probiotics. Many probiotics are derived from milk associated organisms that have been selected to provide fitness benefits in infancy and childhood. Benefits to the host may also be the result of selection arising from microbe-microbe competition. Certain probiotic strains produce anti-pathogen bacteriocins and monopolize space and nutrients, thereby preventing colonization by pathogens.
Earlier this month I attended the 2016 Probiota Americas and International Probiotic Association World Congress in Chicago. One of the standout lectures was on the topic of probiotics in medicine and critical care. Robert Martindale, Chief of General Surgery at Oregon Health & Science University, reviewed findings relevant to our question about probiotics. Here are a few of the studies he highlighted plus some others that he left out:
This study involved 231 Finnish children aged 2–7. The design was a double blind, randomized placebo-controlled trial with Lactobacillus rhamnosus GG. Outcomes were preschool children’s antibiotic use and antibiotic-associated gastrointestinal complaints. The main finding was a significant decrease in gastrointestinal complaints. The investigators also reported reduced antibiotic use in the probiotic group during the 3 year follow up period, suggesting that probiotics might have a durable effect that protects against infections.
This was a systematic review that included 189 studies on probiotics and pregnancy outcomes. The authors concluded that probiotics significantly reduce the risk of gestational diabetes and maternal fasting glucose and may reduce the risk of preeclampsia. (Caveat: a future large trial is needed to validate these findings because this review included small studies with variable study designs)
This systematic review included 21 randomized controlled trials (RCTs) on primary prevention of C. difficile using a variety of probiotics: Saccharomyces boulardii, Lactobacillus casei DN114001, a mixture of L. acidophilus and Bifidobacterium bifidum, and a mixture of L. acidophilus, L. casei and L. rhamnosus. The bottom line: The authors found a significant benefit of probiotics for primary prevention.
The study also examined 4 RCTs examining secondary prevention of C. difficile relapse. No benefit was seen for secondary prevention.
(Bonus: TheNNT.com website reports the number needed to treat is 25 for primary C. difficile prevention and make the recommendation in favor of probiotics).
This recent randomized controlled trial included 235 critically ill participants. The main finding: probiotic use was associated with a significant reduction in incidence of ventilator-associated pneumonia (VAP) (36.4 vs. 50.4 %, p = 0.031) and also longer time before patients developed VAP (p = 0.022).
This review included more than 11,000 participants pooled from 82 studies. They reported a significant reduction in antibiotic associated diarrhea (p< .001). The number needed to treat to obtain benefit is 13, according to the authors of this study.
6. Probiotics for acute infectious diarrhea Allen et al.Cochrane Database Syst Rev. 2010 Nov 10;(11):CD003048. Review. PubMed PMID: 21069673.
This systematic review included 63 RCTs involving children and adults with acute infectious diarrhea. Their main finding: Probiotics decreased mean duration of diarrhea by more than a day (95% CI 16–34 hours) and also reduced stool frequency (95% CI 0.5–1.1 stools). Probiotics also decreased diarrhea lasting >4 days from 45% to 19% (NNT=4).(Analysis courtesy of TheNNT.com)
7. A Meta-Analysis of Probiotic and Synbiotic Use in Elective Surgery: Does Nutrition Modulation of the Gut Microbiome Improve Clinical Outcome? Kinross JPEN J Parenter Enteral Nutr. 2013
This meta-analysis included 13 randomized controlled trials although only 4 were double blinded. Their main finding: probiotics reduce the incidence of postoperative sepsis in elective general surgery:
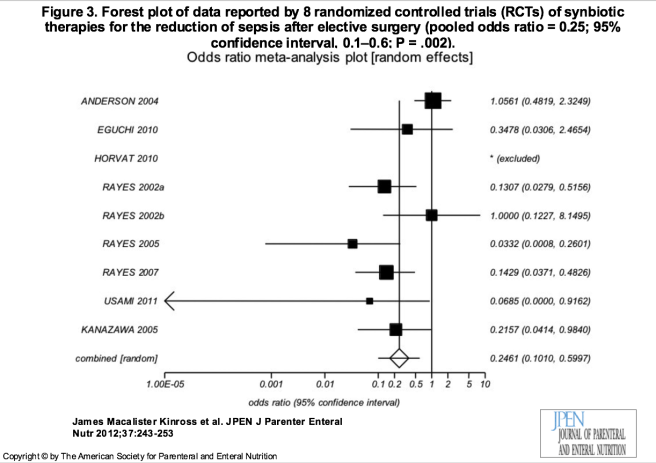
The double blind RCT compared placebo or a formulation containing Lactobacillus acidophilus, L. plantarum, Bifidobacterium lactis and Saccharomyces boulardii in patients undergoing colorectal surgery. The study was stopped early for efficacy. The probiotic group had fewer overall complications, including fewer infections and pneumonia.

From Kotzampassi et al. World J Surg 2015
In addition to these randomized trials and systematic reviews, ongoing research is currently studying whether probiotics reduce viral shedding and duration of symptoms, and whether probiotics can prevent Type 1 diabetes. All these look promising and I will post updates when I have them.
I have a few concluding thoughts about this area of research. First, probiotics are not all the same thing, and even for the same species, much strain specificity exists. Second, there have not been nearly enough probiotic human trials. Financial and regulatory issues have been barriers to large scale trials that may help doctors decide when probiotics can be used effectively and safely. Third, patient selection might be very important. For instance, Some evidence points toward sex differences in response to microbes, including possibly probiotics. Also certain probiotics that are benign for most people might cause problems in immunocompromised persons. Finally, at least one study, the PROPATRIA trial, showed harm (increased deaths) in the probiotic-receiving group. However, that trial had some very significant limitations which we will save for a later post, and a single negative trial should not be a roadblock to future research of probiotics in the critically ill.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
2 replies ›