
The Greek god Janus
Recent discoveries have highlighted the importance of the human microbiome to human health in 1) shaping normal gastrointestinal and immune development, 2) regulation of healthy body weight, 3) prevention of gut and other infections, 4) providing necessary vitamins, 5) influencing mood and normal brain function. These insights have led some researchers to propose that the human microbiota is a forgotten organ, as complex and integrated into the healthy functioning of our bodies as our own organs. For instance, the collective metabolism of microbes in our guts is equivalent to the metabolism carried out by the liver. The microbiota also generates hormones and neurochemicals, with body-wide effects and so might be considered an endocrine organ. However, this microbial organ is unique in containing multitudes of distinct genomes, each with competing genetic interests and potential for conflict. Because of this difference, the microbiome is an organ like no other.
One way the microbiome is a different kind of organ is illustrated by germ free animals. Experimenters are able to raise germ-free animals in a microbe free facility. And, they don’t drop dead (the animals, not the experimenters)! In fact, germ free animals have been reported to live longer than their conventional counterparts. This is true for rats, and in fruit flies, (but perhaps not always)
The only other organs that can be removed thereby increasing longevity are the reproductive organs, but that has a pretty big negative effect on fitness.
The other feature of the microbiome is its potential to turn on you if conditions get bad. Under stress, previously benign bacteria transform into deadly pathogens. This is particularly true of microbes with pathogenic potential, like Pseudomonas and other gram negative bacteria. But even nonpathogens become invasive if given the chance. Finally, microbes from the microbiome immediately disseminate throughout your body after you die, highlighting the fact that the body must expend much effort to corral the microbiome to the reservoirs where it is tolerated.
The Jekyll and Hyde nature of the microbiome explains a variety of phenomena that are otherwise hard to fathom. First, it may explain why the body responds so differently to different foods, for instance to different kinds of fat with the same calorie content. It may also explain why gene expression in humans appears to be exquisitely sensitive to the composition of the microbiota in the gut. It can be inferred that the composition of the microbiota, and the behaviors and functions of the microbiota, have had a profound fitness impact on their hosts during the evolution of life on this planet. Because these impacts are good and bad, our bodies often respond in radically different ways to different microbes, and in opposite ways to nutrients that shape the microbiota.
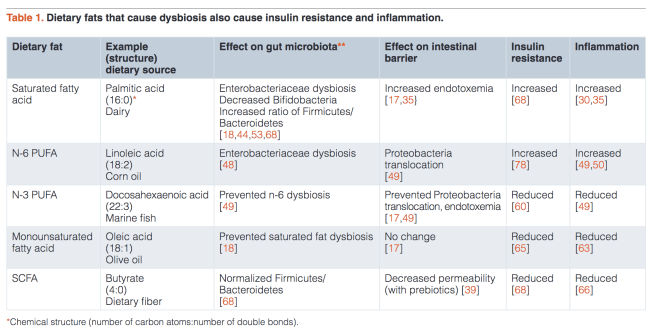
The table above, from (Alcock and Lin 2015) shows the diverging effects of various fats on insulin resistance (a prediabetic state) and inflammation. These differences only make sense if the microbiome is not uniformly mutualistic, instead varying along a mutualism-parasitism continuum.
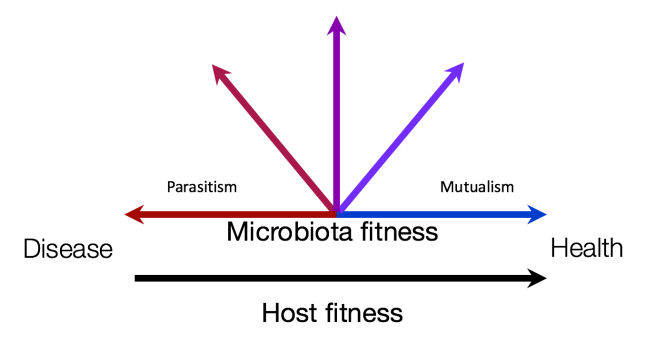
A more realistic view of the microbiome is illustrated above (credit: C. Maley). The fitness impacts of the microbiota range on a spectrum of parastitism (conflict) to mutualism (cooperation). The host is highly sensitive to changes in the microbiome and often responds in a way that compensates for these changes. The combination of host response and microbiome behavior can either lead to health or disease, depending in large part on whether the fitness interests of host and microbiome are aligned or not.
As we described in a recent post, whether cooperative behaviors or conflict exists between human host and microbome often depends on what we eat.
Read our recent paper here: Wasielewski H, Alcock J, Aktipis A.Resource conflict and cooperation between human host and gut microbiota: Implications for nutrition and health. Annals of the New York Academy of Sciences. June 6 2016
Also:
Copyright © Joe Alcock MD
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine

2 replies ›