During the Covid-19 pandemic, many have written about the immune system turning against us.
Ali Daneshkhah, a researcher who published a recent article about Vitamin D and COVID stated: “what seems to kill a majority of COVID-19 patients (is) not the destruction of the lungs by the virus itself. It is the complications from the misdirected fire from the immune system.”
And in Science magazine, esteemed virologist Peter Piot said of COVID-19: It’s a result of your immune defense going into overdrive. Many people do not die from the tissue damage caused by the virus, but from the exaggerated response of their immune system.
Whether the immune system causes misdirected fire in COVID-19 is an open question, although the Kawasaki-like disease caused by COVID-19 seems to be the real deal. But we do know that friendly fire from the immune system is a real, and sometimes lethal problem.
The Jarisch Herxheimer (JH) reaction is one example. The JH reaction or response was named after two dermatologists in 1902 who noticed worsening skin rashes in patients with syphilis who were treated with mercury compounds. When penicillin became the treatment of choice for syphilis, the JH reaction included worsening rash and skin lesions, but also fever, rigors, dangerously low blood pressure, and sometimes death. The JH reaction has been reported in other spirochete infections, including Lyme disease and relapsing fever. This immune-mediated response of JH has been attributed to a sudden increase in pro-inflammatory cytokines, including tumor necrosis factor (TNF).
Borrelia spirochetes remodel their outer surface lipoproteins when they transition from ticks to humans; these changes help the microbes avoid detection from their host, acting like an invisibility cloak. On the other side, hosts have been under selection to become very good at detecting subtle cues of pathogens that can trigger an immune response that clears the infection. When penicillin is given for syphilis, the microbial surface undergoes changes, revealing molecular patterns that initiate antibody and complement-driven immune responses. In this way, antibiotics can suddenly reveal widespread infection to the immune system. For patients with the JH reaction, antibiotics can tip the contest between pathogen and host too far towards host immunity, sometimes unleashing a devastating, fatal immune response.
Auto-immunity is another example of immune responses that are out of control.
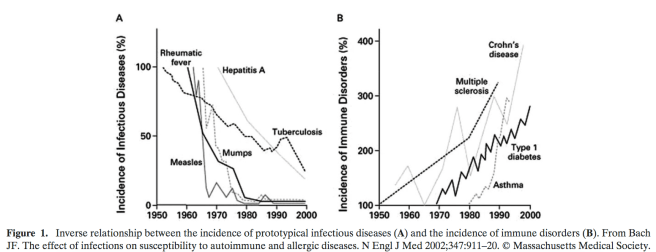
Over the last half-century, autoimmune-related disorders have increased (e.g. multiple sclerosis, Crohn disease, asthma, and Type I diabetes) at the same time that the incidence of many infectious diseases has decreased dramatically (e.g. measles, tuberculosis, rheumatic fever, and hepatitis A) while that for (Bach 2002). The so-called hygiene hypothesis gained attention with the identification of an inverse relationship between family size and allergic diseases and suggested that childhood infections have a protective effect for allergy. Also termed the “old friends” hypothesis, it contends that increases in autoimmune inflammatory disorders in developed countries are partly attributable to decreasing exposure to microorganisms with which humans coevolved. Here too,antibiotics are part of the problem, decreasing the diversity of gut microbes that babies and children are exposed to. Other changes, like proximity to animals, also affects allergy risk.
A 2015 study by Stein and colleagues in the New England Journal of Medicine showed differences in asthma risk between genetically similar Amish and Hutterite children. 1 in 20 Amish kids get asthma, while 1 in 5 Hutterite children have asthma. The difference could be explained by childhood exposure to farm animals (and microbes). The main difference between these two farming groups is their exposure to farm animals. Amish have single family dairy farms and live in close proximity to their animals. By comparison, the Hutterites farm on communal highly industrialized farms. That difference may be enough to protect Amish children from allergies.
What is the agent that protects Amish kids from asthma? Microbes. Amish farming lifestyle makes them have relatively greater exposure to farm animal microbes and antigens.
In this respect the Amish are like the Karelians, a Russian group of traditional farmers with a markedly lower allergy risk compared to genetically similar Finns.

So these associations are not exactly new. What is different about this study is that they showed that the Amish children develop their immune systems differently, with more neutrophils and fewer eosinophils. Neutrophils are innate immune cells that chiefly fight bacterial infections, while eosinophils are immune cells responsible for defense against helminths and are the cells responsible for type 1 hypersensitivity reactions, like asthma. The gene expression patterns of white blood cells in Amish vs Hutterite were also markedly different:

Circulating immune cells had different gene expression patterns in the Amish (blue) compared to the Hutterites (red). These differences are developmental and entirely driven by differences in microbial exposure. Remember, Amish and Hutterite have almost the same ancestors, and are thus remarkably genetically similar. So all these differences, in immune cells and in asthma phenotype, are essentially environmental. The dramatic differences in gene expression seen above is a good example of a human reaction norm (discussed in previous posts like this one). I propose that these changes in immune cell development, and the responses to microbial signals described in the Stein paper, evolved because they were adaptive in historic environments. This idea is similar to the hygiene hypothesis, which uses evolutionary principles, including gene environment mismatch to explain why more autoimmune diseases occur when microbes and pathogens are absent.
What we don’t know is what kind of microbial exposures, and which route, are most important in determining asthma and allergy risk. In the NEJM study, showed that protection from airway hypersensitivity could be achieved when dust from Amish homes was instilled into the noses of experimental mice. So maybe nasal inoculation of house dust is the most important route exposure. That makes sense since airway exposure would be expected to affect the risk of asthma, an airway disease. But other studies suggest the oral and gut route are important. For instance, Hesselmar and colleagues have looked at pacifier cleaning methods in relation to allergy:

Kids whose parents “clean” their pacifiers by sticking it in their mouth first had fewer allergies, even more so if the child was also born vaginally instead of by c-section.
Can we draw any lessons from immune overreactions that can be explained by the hygiene hypothesis and in the Jarisch-Herxheimer reaction? Yes. The immune system does not typically over-react under ordinary circumstances – for instance those contexts and environments that humans evolved in. It takes special circumstances for these overreactions to occur. Some novel exposure, like antibiotics, and c-section birth, and decreased exposure to diverse microbes or to animals (domesticated or wild) is necessary to drive JH reactions or hygiene-related autoimmune diseases.
How about COVID-19? As we suggested in the last entry, having a low diversity microbiome might alter immunity and result in an impaired anti-viral response. It is also possible that the novelty of this coronavirus may explain a possibly excessive immune response. Another possibility is that the response is not excessive at all (on average). The smoke detector principle or the immune brinksmanship concepts may be sufficient to explain some seemingly excessive responses. We will talk more about this later this week.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine
Leave a comment