We are going to continue our conversation about diet and diabetes, and hypotheses about the adaptive value of insulin resistance. Before Tuesday’s class, lets review what insulin does and what insulin resistance is.
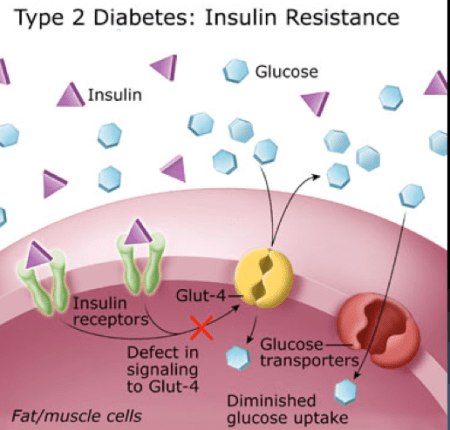
Insulin is a hormone that regulates energy storage and glucose transport in particular. Insulin activates insulin receptors which then acts on the GLUT 4 cell membrane glucose transporter. Under ordinary circumstances, insulin allows the GLUT 4 transporter to move glucose into muscle and fat cells. Insulin promotes the storage of glucose as glycogen in the liver and skeletal muscle, and insulin increases the storage of energy in fatty tissue, by increasing triglyceride movement into adipocytes. Insulin resistance, by contrast, decreases cellular energy uptake in most somatic tissues, and antagonizes “anabolic” glycogen and triglyceride storage.
During insulin resistance (IR), the Insulin receptor signaling to the GLUT 4 is inhibited, preventing GLUT 4 from transporting glucose. Thus, in IR, glucose is prevented from entering muscle and fat cells.
Although there is writing assignment this week, I want students to read this Odegaard and Chawla 2013 paper and be prepared to have a discussion about insulin resistance. Read on:
In a recent January issue of Science, Odegaard and Chawla (2013) reviewed the varied effects of insulin resistance and inflammation. Refreshingly, they take an evolutionary approach. They write: “An appreciation of the adaptive context in which these responses arose is useful for understanding their pathogenic actions in disease.” This statement articulates the evolutionary basis for health and disease – the raison d’être of evolutionary medicine – in a journal as prestigious as Science, no less.

In common with another recent review by Tsatsoulis et al. (click here to read their review on insulin resistance), Odegaard and Chawla point out the linkages between metabolic disturbances and obesity, citing the pro-inflammatory effects of excess fat as a major source of unexplained mortality. In parallel with Tsatsoulis et al.’s, arguments, Odegaard and Chawla write:
“Specifically, we present the argument that key mediators of obesity-induced metabolic disease, such as insulin resistance and inflammation, are evolutionarily conserved adaptive traits with maladaptive effects in the modern obesogenic environment.”
Normal physiologic conditions demonstrate the function of insulin resistance in re-allocating energetic resources.
Odegaard and Chalwa write:
“For example, both infection and pregnancy require organisms to reserve priority nutrient access for an emerging metabolic requirement—immune system activation and fetal development, respectively, in this instance. Organisms meet these requirements by decreasing systemic insulin sensitivity (developing insulin resistance), decreasing nutrient uptake by nonpriority tissues and reserving glucose for priority cells.”
Odegaard and Chalwa explain the adaptive value of IR during sepsis, hibernation, and pregnancy:
“Indeed, insulin resistance confers evolutionary advantages and enhances organismal fitness in each of these categories: fueling immune function to combat infection, switching hibernator metabolic substrate preference from glucose to lipids to avoid starvation, and reserving metabolic resources for fetal development to optimize reproduction.”
For Odegaard and Chalwa, obesity provides the example of maladaptive IR. They make much the same argument as the Tsatsoulis et al. about the toxic effects of fat and adipocyte’s limited ability to absorb excessive energy from the diet:
“After the adipose tissue depots collapse, skeletal myocyte and hepatocyte depots would similarly fail, followed closely by nonprofessional nutrient-storage tissues. Organisms would be able to quite literally eat themselves to death.”
Both sets of authors cite pro-inflammatory lipid intermediaries, including ceramide and oxidized lipids, as major mediators of adipocyte inflammation and pathological dysregulation that leads to inflammatory complications of obesity.
A major question raised by these observations is the problem of energy excess really just a storage problem? Is the human body simply ill-equipped to metabolize and store energy because of some limit to storage capacity?
Again, the observations my colleagues and I raised in our 2012 Quarterly Review of Biology article Nutrient signaling: evolutionary origins of the immune-modulating effects of dietary fat are instructive: e.g. not all fats are created equal with regard to inflammation and insulin signaling. Despite being equipotent energetically, saturated fats and omega-3 fatty acids have radically different immune effects. Unlike many saturated fats, omega-3 fat does not promote obesity, nor does it result in pro-inflammatory signaling. Omega-3 fats also antagonize insulin resistance.
One of the mechanisms by which fats are thought to elicit IR is by activating serine kinases, such as JNK. Notably, JNK was highlighted by both Tsatsoulis et al., and Odegaard and Chawla as a link between nutrient excess, inflammation and insulin resistance. However, as Holzer and colleagues demonstrated, Omega-3 fatty acids and saturated fatty acids have directly opposite effects on JNK serine kinases. These results highlight the fact that energy excess is an incomplete explanation for the problem of IR and inflammation.
The pleiotropic effects of different species of dietary fats argue against a generic limit to the body’s ability to store food energy. Perhaps something else is going on here?
References:
Alcock J, Franklin ML, Kuzawa CW. Nutrient signaling: evolutionary origins of the immune-modulating effects of dietary fat. Q Rev Biol. 2012 Sep;87(3):187-223.
Holzer R. G., Park E.J., Li N., Tran H., Chen M., Choi C., Solinas G., Karin M. 2011. Saturated fatty acids induce c-Src clustering within membrane sub- domains, leading to JNK activation. Cell 147:173– 184.
Odegaard JI, Chawla A. Pleiotropic Actions of Insulin Resistance and Inflammation in Metabolic Homeostasis. Science 339, 172 (2013); doi: 10.1126/science.1230721
Tsatsoulis A, Mantzaris MD, Bellou S, Andrikoula M.Insulin resistance: An adaptive mechanism becomes maladaptive in the current environment – An evolutionary perspective. Metabolism. 2012 Dec 18. pii: S0026-0495(12)00438-6. doi: 10.1016/j.metabol.2012.11.004.
Categories: Uncategorized
Joe Alcock
Emergency Physician, Educator, Researcher, interested in the microbiome, evolution, and medicine

Leave a comment